Hi CUET aspirants, Welcome to Amans Maths Blogs (AMBIPi). In this post, you will get CUET Chemistry Mock Aldeydes Ketone Carboxylic Acids AMBIPi. This CUET Chemistry Mock Test is of the Chemistry chapter Aldeydes Ketone Carboxylic Acids. Before solving these CUET Chemistry questions, you must read CUET Chemistry Notes, which helps you to revise CUET Chemistry Syllabus and then you must need to solve CUET Previous Years Questions Papers.
CUET Chemistry Mock Test
CUET Chemistry Question No: 1
Carbonyl compounds are the constituents of
Option A : fabrics
Option B : flavouring
Option C : plastics and drugs
Option D : All of these
Show/Hide Answer Key
Option D: All of these
Carbonyl compounds are of almost importance to organic chemistry.They are the constituents of fabrics, flavourings, plastics and drugs.
CUET Exam Chemistry Question No: 2
The most suitable reagent for the conversion of R — CH2 — OH → R — CHO is
Option A : KMnO4
Option B : K2Cr2O7
Option C : H2SO4
Option D : PCC (pyridinium chlorochromate)
Show/Hide Answer Key
Option D: PCC (pyridinium chlorochromate)
The most suitable reagent for the conversion of alcohol
(RCH2 — OH) to aldehyde ( R — CHO ) is PCC:
R — CH2OH → R — CHO Pyridinium chlorochromate is a mild oxidising agent which causes the conversion of alcohol to aldehyde and not carboxylic acid. Whereas, K2Cr2O7 and KMnO4 are strong oxidising agents which causes conversion of alcohol directly to acid and H2SO4 is a dehydrating agent. Thus, they cannot be used for the conversion.
CUET UG Chemistry Question No: 3
Alcohol vapours are passed over which of the following catalysts to give aldehydes and ketones?
Option A : S or Pd
Option B : Ag or Cu
Option C : F or Cl
Option D : Li or K
Show/Hide Answer Key
Option B: Ag or Cu
Alcohol vapours are passed over heavy metal catalysts (Ag or Cu) to give aldehydes and ketones. Primary and secondary alcohols give aldehydes and ketones respectively.
CUET Domain Subject Chemistry Question No: 4
The reaction, RCN + SnCI2 + HCI → RCH = NH→ RCHO is known as
Option A : Etard reaction
Option B : Haloform reaction
Option C : Gattermann-Koch reaction
Option D : Stephen reaction
Show/Hide Answer Key
Option D: Stephen reaction
RCN + SnCI2 + HCI → RCH = NH→ RCHO is known as This reaction is called Stephen reaction, where nitriles are reduced to corresponding imine with stannous chloride in the presence of HCl, which on hydrolysis gives corresponding aldehyde.
CUET BSc Chemistry Question No: 5
Reaction by which benzaldehyde cannot be prepared is
Option A :
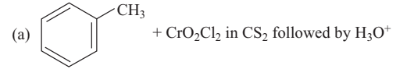
Option B :
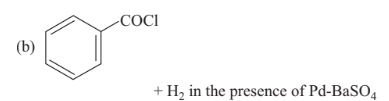
Option C :

Option D :
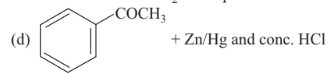
Show/Hide Answer Key
Option D: D
CUET Entrance Exam Chemistry Question No: 6
The compounds methanal, ethanal and propanone are miscible with water because they form
Option A : van der Waals’ forces with water
Option B : ‘H’-bonding with water
Option C : dipole-dipole bond with water
Option D : ion-dipole bond with water
Show/Hide Answer Key
Option B: ‘H’-bonding with water
The lower members of aldehydes and ketones such as methanal, ethanal and propanone are miscible with water in all proportions because they form hydrogen bonding with water.
CUET Exam Question No: 7
Identify the example in which nucleophilic addition followed by elimination reaction occur respectively in the carbonyl compound
Option A : Addition of Grignard reagent
Option B : Addition of sodium sulphide
Option C : Addition of HCN
Option D : Addition of NH3
Show/Hide Answer Key
Option D: Addition of NH3
CUET Chemistry Practice Questions No: 8
Consider the reaction given below.
CH2 = CH — CHO → CH2 = CH — CH2 — OH
Which of the following is the suitable reagent for the conversion of following reaction?
Option A : NaBH4
Option B : Ni/H2
Option C : Zn/Hg/HCI
Option D : Red P + Hi
Show/Hide Answer Key
Option A: NaBH4
The suitable reagent for the given conversion is NaBH4
CH2 = CH — CHO → CH2 = CH — CH2 — OH
It is a selective reagent and does not reduce carbon-carbon double bond. Thus, unsaturated alcohols can be prepared by this reagent.
CUET Chemistry Sample Paper Question No: 9
On heating an aldehyde with Fehling’s reagent, a reddish brown precipitate is obtained due to the formation of
Option A : CuO
Option B : Cu2O
Option C : CuSO4
Option D : Cu2+ + OH–
Show/Hide Answer Key
Option B: Cu2O
On heating an aldehyde with Fehling’s reagent, a reddish
brown precipitate is obtained due to the formation of Cu2O. Aldehydes are oxidised to corresponding carboxylate anion. The reaction involved is as follows
RCHO + 2Cu2+ + 5 OH → RCOO + Cu2O↓ + 3H20
Red brown
ppt.
CUET Chemistry Mock Test Question No: 10
Which of the following compounds produces an orange-red precipitate with 2, 4-DNP reagent?
Option A : Acetamide
Option B : Dimethyl ether
Option C : Butanone
Option D : Propylbutanoate
Show/Hide Answer Key
Option C: Butanone
2, 4-DNP reagent reacts with carbonyl compounds (aldehydes and ketones) to produce orange-red precipitate. Among the given compounds, butanone is a carbonyl compound and, thus can react with 2, 4-DNP reagent.
CUET Chemistry Question No: 11
Predict the products in the given reaction.
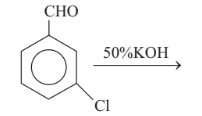
Option A :
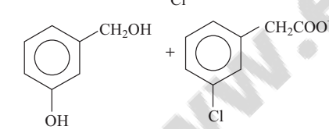
Option B :
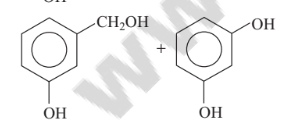
Option C :
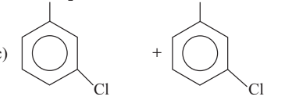
Option D :
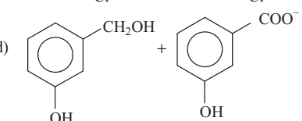
Show/Hide Answer Key
Option C: C
CUET Exam Chemistry Question No: 12
Some higher members of aliphatic carboxylic acids known as fatty acids occur in natural fats as esters of glycerol. How many C-atoms do they contain?
Option A : C12 – C18
Option B : C10 – C15
Option C : C5 , – C10
Option D : C20 – C30
Show/Hide Answer Key
Option A: C12 – C18
(a) Some higher members of aliphatic carboxylic acids containing C12 – C18 are known as fatty acids and they occur in natural fats as esters of glycerol. Therefore, the correct option is (a).
CUET UG Chemistry Question No: 13
What is the common name of 2-methylpropanoic acid?
Option A : Adipic acid
Option B : Crotonic acid
Option C : Isobutyric acid
Option D : Acrylic acid
Show/Hide Answer Key
Option C: Isobutyric acid
CUET Domain Subject Chemistry Question No: 14
Select the acid(s) which cannot be prepared by Grignard reagent?
Option A : Acetic acid
Option B : Succinic acid
Option C : Formic acid
Option D : All of these
Show/Hide Answer Key
Option A: Acetic acid
CUET BSc Chemistry Question No: 15
Carboxylic acids have higher boiling points than aldehydes, ketones and even alcohols of comparable molecular mass. It is due to
Option A : more extensive association of carboxylic acid via
van der Waals’ force of attraction
Option B : formation of carboxylate ion
Option C : formation of intramolecular H-bonding
Option D : formation of intermolecular H-bonding
Show/Hide Answer Key
Option D: formation of intermolecular H-bonding
CUET Entrance Exam Chemistry Question No: 16
Which of the following reagents is/are used for the conversion of ethanoic acid to ethanoic anhydride?
Option A : SOCI2, Δ
Option B : PCI3 , Δ
Option C : P2O5, Δ
Option D : All of these
Show/Hide Answer Key
Option C: P2O5, Δ
Reagents that are used for the conversion of ethanoic acid to ethanoic anhydride are mineral acids such as H2SO4 or with P2O5
CUET Exam Question No: 17
Diborane easily reduce the functional group such as
Option A : acid
Option B : ester
Option C : nitro
Option D : halo
Show/Hide Answer Key
Option A: Acid
Diborane does not easily reduce functional groups such as ester, nitro, halo etc. Carboxylic acids are reduced to primary alcohols by lithium aluminium hydride or with diborane.
CUET Chemistry Practice Questions No: 18
Name the product formed during the decarboxylation of malonic acid.
Option A : Acetic acid
Option B : Ethanone
Option C : Propanone
Option D : Formic acid
Show/Hide Answer Key
Option A: Acetic acid
CUET Chemistry Sample Paper Question No: 19
Aromatic carboxylic acids do not undergo Friedel-Crafts reaction because
Option A : carboxyl group acts as an activating and meta-directing group
Option B : carboxyl group act as a deactivating and ortho and para directing group
Option C : carboxyl group act as an activating and ortho-directing group
Option D : carboxyl group acts as deactivating and the catalyst
Show/Hide Answer Key
Option B: carboxyl group act as a deactivating and ortho and para directing group
The order of reactivity of hydrogen halides with ether is as follows. HI > HBr > HCl. The cleavage of ethers take place with concentrated HI or HBr at high temperature.
CUET Chemistry Mock Test Question No: 20
Which of the following acid is used in rubber, textile, dyeing, leather and electroplating industries?
Option A : Hexanedioic acid
Option B : Ethanoic acid
Option C : Methanoic acid
Option D : Sodium benzoate
Show/Hide Answer Key
Option C: Methanoic acid
