Hi CUET aspirants, Welcome to Amans Maths Blogs (AMBIPi). In this post, you will get CUET Chemistry Notes Study Materials Polymers AMBIPi. This CUET Chemistry Notes are designed by analyzing to the CUET Syllabus and CUET Previous Years Questions Papers.
CUET Chemistry Notes
CUET Chemistry Polymers: Important Points to Remember
There are following important points in this chapter of Polymers.
CUET Chemistry: Polymer
The word ‘polymer’ is coined from two Greek words: poly means many and mer means unit or part.
The term polymer is defined as very large molecules having high molecular mass (103–107u).

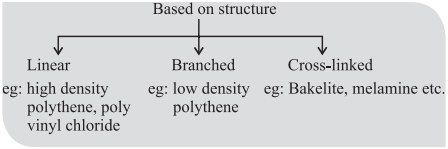
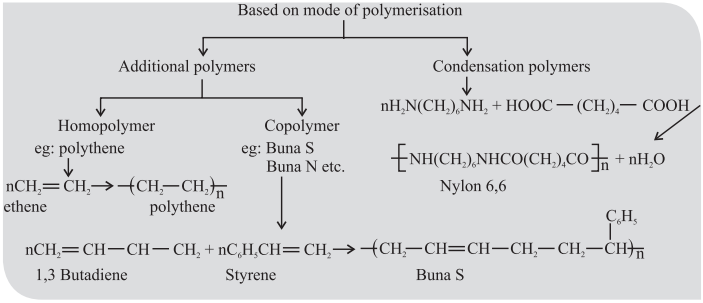
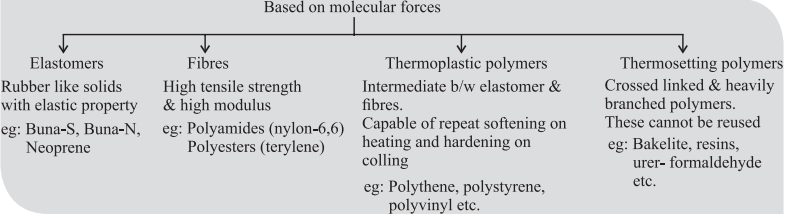
CUET Chemistry: Important Terms
Low density polythene:
It is obtained by the polymerization of ethene under high pressure of 1000 to 2000 atmospheres at a temperature of 350 K to 570 K in the presence of traces of dioxygen or a peroxide initiator (catalyst).
The low density polythene (LDP) obtained through the free radical addition and H-atom abstraction has highly branched structure.
Low density polythene is chemically inert and tough but flexible and a poor conductor of electricity.
High density polythene:
It is formed when addition polymerization of ethene takes place in a hydrocarbon solvent in the presence of a catalyst such as triethylaluminium and titanium tetrachloride (Ziegler-Nata catalyst) at a temperature of 333 K to 343 K and under a pressure of 6-7 atmospheres.
High density polythene (HDP) thus produced, consists of linear molecules and has a high density due to close packing.
It is also chemically inert and more tougher and harder.
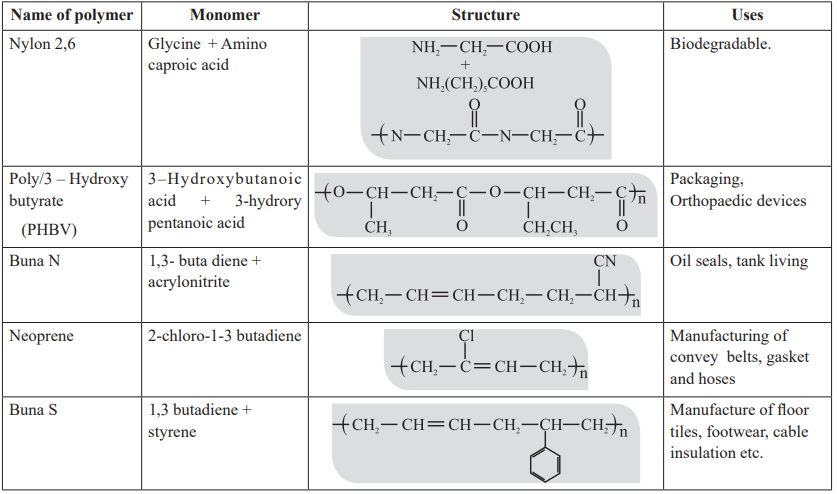
Biodegradable polymers:
Aliphatic polyesters are one of the important classes of biodegradable polymers. Some important examples are given below:
Poly β-hydroxybutyrate – co-β-hydroxy valerate (PHBV)
PHBV is used in speciality packaging, orthopaedic devices and in controlled release of drugs.
PHBV undergoes bacterial degradation in the environment.
Number Average Molecular Mass i.e.
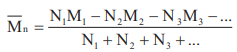 ,
,
Where N1, N2, N3 etc are the number of macromolecules with molecular masses M1, M2, M3 etc. respectively.
Weight average molecular mass i.e.,
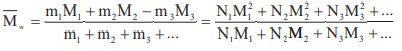
where N1, N2, N3 …. etc. are the number m1, m2, m3 are the masses of macromolecules with molecular masses M1, M2,M3 of respectively.
Poly Dispersity index (PDI): PDI = Mw / Mn.
Natural polymers are also called monodisperse polymers since their molecules have same or a narrow range of molecular masses for natrual polymers PDI = 1.
Synthetic polymers are also called poly disperse polymers. Since their molecules have a wide range of molecular masses. For synthetic polymers, PDI > 1.
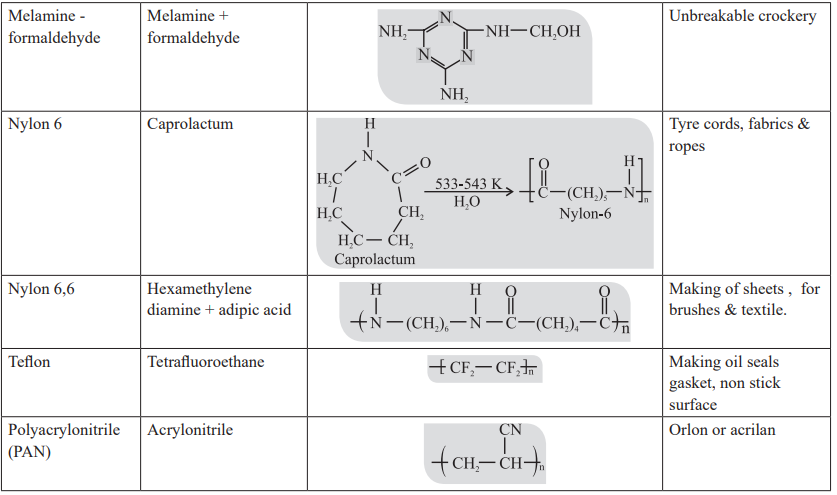
CUET Chemistry: Examples of Polymer
Strength of forces present in different polymers elastomer < thermoplastic < fibres < thermosetting.
CUET Chemistry Mock Test
Now, you have revised the this CUET Chemistry chapter. So, you must need to practice CUET Chemistry Sample Papers. By solving these CUET Chemistry questions, you will be more confident about your CUET preparations.
